Cohesin helps maintain cell wall in budding yeast cells
– Anoushka Dasgupta
Mutations in proteins needed to maintain the structure of the cell wall in yeast cells may weaken it, threatening the cell’s survival. A study by researchers at the Department of Biochemistry, published in Genetics, has explored the role of one such protein complex called cohesin.
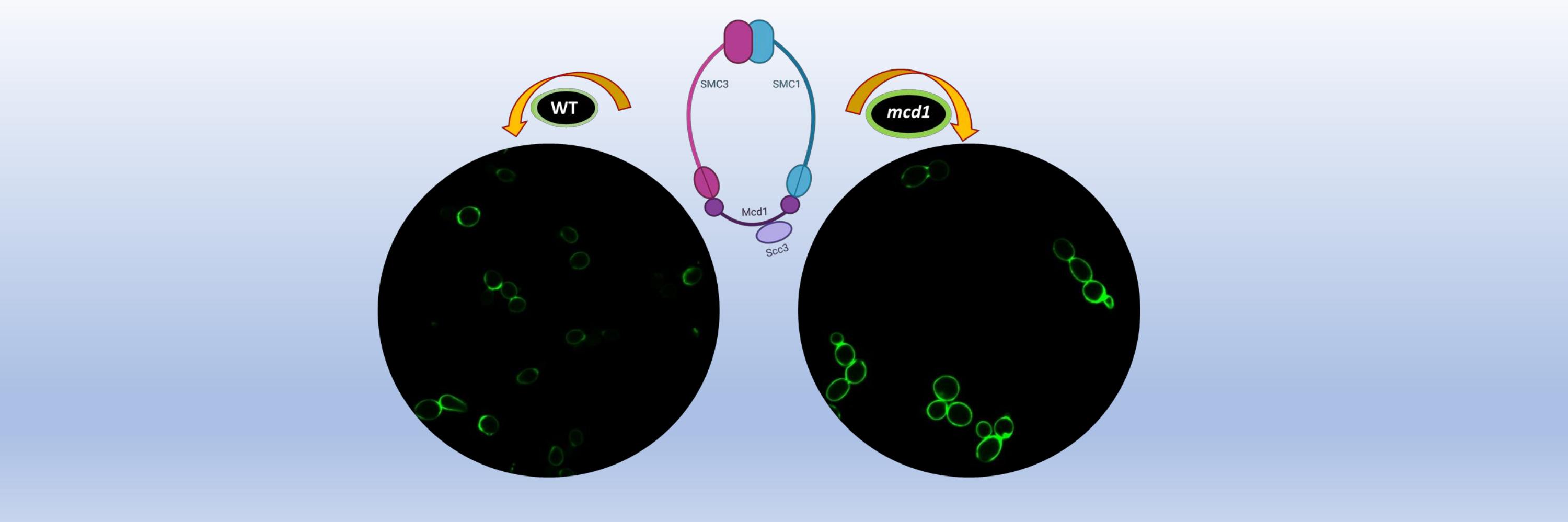
Cohesins attach to various parts of the cell’s chromosomes and help organise and distribute genetic material during cell division. A previous study by the team, published in PNAS, described a unique mechanism by which cohesin represses the expression of subtelomeric genes, even when the genes are present further away from sites known to be silenced by the SIR-complex, which contains Sir2, a well-established conserved silencing regulator.
In the new study, the researchers found that yeast cells with mutations in cohesin proteins had distinctive characteristics. Their cell walls were resistant to an enzyme called zymolyase, and showed large depositions of chitin, known to fortify cell walls. They were also more sensitive to chemical inducers of cell wall stress and to elevated temperature; interestingly the temperature sensitivity could be remedied by altering the osmotic stress. This hinted at the presence of cell wall defects. A compensatory stress signalling pathway is activated in these mutants, which is important for their survival under elevated cell wall stress conditions. Preferential deregulation of expression of multiple cell wall genes was seen in the cohesin mutants, which may result in the observed cell wall defects. Cohesins, therefore, are vital for regulating both chromosome segregation during cell division and cell wall maintenance, independently.
References:
Kothiwal D, Gopinath S and Laloraya S, Cohesin dysfunction results in cell wall defects in budding yeast, 14 December 2020, Genetics, iyaa023 (Advance online publication)
https://doi.org/10.1093/genetics/iyaa023
Kothiwal D and Laloraya S, A SIR-independent role for cohesin in subtelomeric silencing and organization, Proc Natl Acad Sci USA, 2019 Mar 19;116(12):5659-5664.
doi: 10.1073/pnas.1816582116
Lab website:
https://biochem.iisc.ac.in/shikha-laloraya.php




