PACEMAKER CHANNELS REGULATE THE BRAIN’S LOCAL FIELD POTENTIAL
Action potentials, or spikes, are specific, transient changes in the electrical potential of the membrane of a neuron, and play a crucial role communication among neurons. However, these are not the only electrical signals generated in the brain. Researchers have long known about another class of electrical signals called the local field potential (LFP), which refers to the slowly-varying component of the extracellular potential in a particular brain region. Although traditional dogma describes LFPs as a composite signal that reflects inputs impinging on neurons in that brain region, the precise origins of LFPs and how they are regulated are yet to be fully understood.
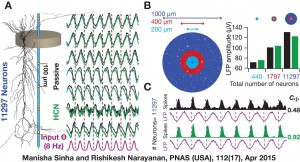 A recent study by ManishaSinha and Rishikesh Narayanan furthers our understanding of LFPs by showing that these electrical signals are not mere reflections of neuronal inputs, but are critically regulated by “subthreshold ion channels” that are present in neuronal dendrites.*Ion channels are pores in the neuronal membrane that regulate the flow of ions between the exterior and interior of neurons, thus mediating the generation of neuronal spikes. “Our study uncovers a distinct role for neuronal pacemaker channels formally referred to as the hyperpolarization-activated cyclic nucleotide-gated (HCN) channels, in regulating LFPs,” says Narayanan. HCN channels allow positively charged ions to enter neurons and, uniquely, are “open” at voltages below the threshold for action potentials.
A recent study by ManishaSinha and Rishikesh Narayanan furthers our understanding of LFPs by showing that these electrical signals are not mere reflections of neuronal inputs, but are critically regulated by “subthreshold ion channels” that are present in neuronal dendrites.*Ion channels are pores in the neuronal membrane that regulate the flow of ions between the exterior and interior of neurons, thus mediating the generation of neuronal spikes. “Our study uncovers a distinct role for neuronal pacemaker channels formally referred to as the hyperpolarization-activated cyclic nucleotide-gated (HCN) channels, in regulating LFPs,” says Narayanan. HCN channels allow positively charged ions to enter neurons and, uniquely, are “open” at voltages below the threshold for action potentials.
A recent study by ManishaSinha and Rishikesh Narayanan furthers our understanding of LFPs by showing that these electrical signals are not mere reflections of neuronal inputs, but are critically regulated by “subthreshold ion channels” that are present in neuronal dendrites
“To understand how these subthreshold-activated channels modulate the LFP, we constructed thousands of realistic model neurons, stimulated them with low-frequency inputs and measured the LFP,” explains Narayanan. His team found that introducing HCN channels in the model neurons resulted in a phase-lead in the LFP. The phase of the spikes, on the other hand, lagged when HCN channels were present, relative to the spike signal recorded without those channels. Along with alteration of the phase properties of spikes and LFP, the activity of HCN channels also increased the coherence between the phases of the two types of signals.
*ManishaSinha and Rishikesh Narayanan. 2015. HCN channels enhance spike phase coherence and regulate the phase of spikes and lfpsin the theta-frequency range. Proceedings of the National Academy of Sciences (USA). 112(17): E2207-E2216.




 A recent study by ManishaSinha and Rishikesh Narayanan furthers our understanding of LFPs by showing that these electrical signals are not mere reflections of neuronal inputs, but are critically regulated by “subthreshold ion channels” that are present in neuronal dendrites.*Ion channels are pores in the neuronal membrane that regulate the flow of ions between the exterior and interior of neurons, thus mediating the generation of neuronal spikes. “Our study uncovers a distinct role for neuronal pacemaker channels formally referred to as the hyperpolarization-activated cyclic nucleotide-gated (HCN) channels, in regulating LFPs,” says Narayanan. HCN channels allow positively charged ions to enter neurons and, uniquely, are “open” at voltages below the threshold for action potentials.
A recent study by ManishaSinha and Rishikesh Narayanan furthers our understanding of LFPs by showing that these electrical signals are not mere reflections of neuronal inputs, but are critically regulated by “subthreshold ion channels” that are present in neuronal dendrites.*Ion channels are pores in the neuronal membrane that regulate the flow of ions between the exterior and interior of neurons, thus mediating the generation of neuronal spikes. “Our study uncovers a distinct role for neuronal pacemaker channels formally referred to as the hyperpolarization-activated cyclic nucleotide-gated (HCN) channels, in regulating LFPs,” says Narayanan. HCN channels allow positively charged ions to enter neurons and, uniquely, are “open” at voltages below the threshold for action potentials.