The Curious Case of Dynein: Regulation by Myosin I
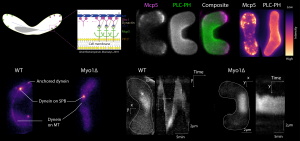
Molecular motors are tiny machines that convert the energy from the breakdown of ATP to mechanical work and hence bring about changes in the organisation of cellular components, including cargo, organelles and nuclear material. There are three major classes of motor proteins that have been identified: (i) myosins, which are associated with cellular polymers called filamentous actin, and (ii) kinesins and (iii) cytoplasmic dynein, which are associated with pipe-like structures called microtubules. Cell division requires the precise orchestration of a number of proteins in the cell, primarily the microtubules and molecular motors. Cytoplasmic dynein is especially important during cell division for ensuring that the nuclear material is organised properly. In this study, we have uncovered that the localisation and activity of dynein during meiotic cell division in fission yeast is dependent on myosin I. This discovery highlights one of the few examples of interplay between actin-based and microtubule-based motors inside the cell. Studies such as these are important for understanding division of not just healthy cells, but also in cases when the cell division machinery goes rogue, such as during cancer.
Reference: Fission yeast myosin I facilitates PI(4,5)P2–mediated anchoring of cytoplasmic dynein to the cortex. Jerrin M. Thankachan, Stephen S.
Nuthalapati, Nireekshit A. Tirumala, and Vaishnavi Ananthanarayanan. Proceedings of the National Academy of Sciences 2017 (DOI: 10.1073/pnas.1615883114)
Website: www.be.iisc.ernet.in/~vaish




