Mtb fitness relies on sulphur metabolism
The metabolism of two amino acids – cysteine and methionine – is critical for the survival of the most successful human pathogen, Mycobacterium tuberculosis (Mtb).Cysteine functions as a biosynthetic precursor for essential cofactors such as Fe-S clusters and a primary antioxidant buffer, mycothiol. Fe-S cluster proteins and mycothiol coordinate Mtb’s ability to respire, resist oxidative stress, counteract anti-TB drugs, and establish infection. Therefore, it is essential to understand the pathways that Mtb utilises to generate cysteine and its subsequent utilisation to produce Fe-S clusters and mycothiol.
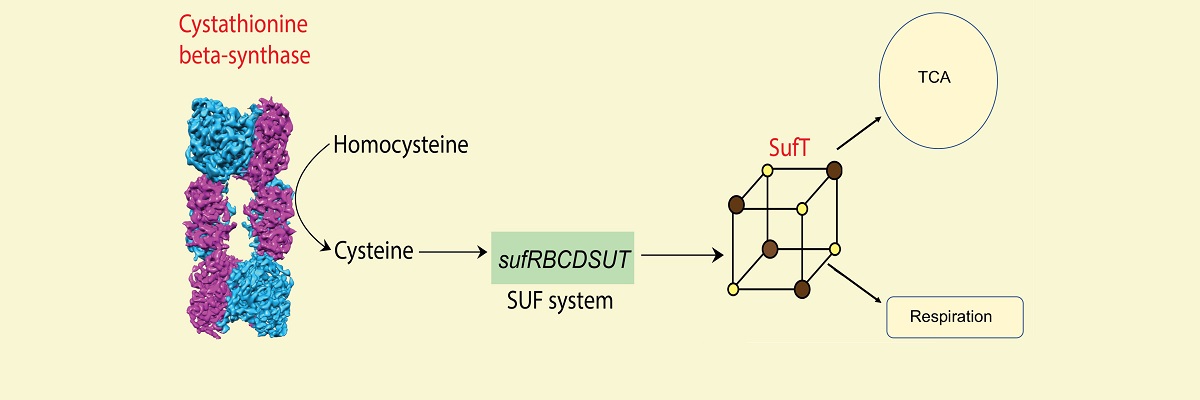
Researchers led by Amit Singh, Associate Professor at the Department of Microbiology and Cell Biology, have now discovered that Mtb generates cysteine via the reverse transsulfuration pathway (RTS). They have biochemically, biophysically, and genetically characterised the first rate-limiting enzyme of the RTS pathway, called cystathionine beta-synthase (Cbs).
An important aspect of this work was done in collaboration with Somnath Dutta, Assistant Professor at the Molecular Biophysics Unit at IISc and Sunil Laxman, Associate Investigator at NCBS/inStem. Dutta’s group characterised Cbs using biophysical techniques, transmission electron microscopy and single-particle cryo-electron microscopy (cryo-EM)-based atomic model determination of tetrameric assembly of native and S-adenosyl methionine (SAM)-bound Mtb Cbs, which provided unprecedented insight into the mechanism of allosteric activation of Mtb Cbs. In collaboration with Laxman, the team discovered that SAM depletion results in the degradation of Cbs, which triggers a non-canonical pathway for methionine biosynthesis in Mtb. Inhibition of RTS and the non-canonical methionine biosynthesis pathway killed Mtb by inducing oxidative stress. Lastly, Cbs is essential for Mtb to cause infection during HIV-TB coinfection. These findings were published in Science Advances.
In another study, the researchers identified the Fe-S cluster biogenesis machinery in Mtb and showed its requirement for mycobacterial respiration, metabolism, and survival in mice. This work was published in PloS Pathogens.
Altogether, the RTS pathway and Fe-S cluster biogenesis represent major metabolic pathways necessary for Mtb’s survival and ability to cause infection. This work has generated new knowledge to understand the intricacies of sulphur metabolism in Mtb, which can be useful for therapeutic applications.
REFERENCES:
Bandyopadhyay P, Pramanick I, Biswas R, Sabarinath PS, Sreedharan S, Singh S, Rajmani RS, Laxman S, Dutta S, Singh A, S-Adenosylmethionine–responsive cystathionine β-synthase modulates sulfur metabolism and redox balance in Mycobacterium tuberculosis, Science Advances (2022).
https://www.science.org/doi/10.1126/sciadv.abo0097
Tripathi A, Anand K, Das M, O’Niel RA, Sabarinath PS, Thakur C, Reddy RLR, Rajmani RS, Chandra N, Laxman S, Singh A, Mycobacterium tuberculosis requires SufT for Fe-S cluster maturation, metabolism, and survival in vivo, PloS Pathogens (2022).
https://pubmed.ncbi.nlm.nih.gov/35427399/
LAB WEBSITES:
https://mcbl.iisc.ac.in/amit-singh/
http://cidr.iisc.ac.in/amit/
https://somnath92.wixsite.com/website










