Orbital Engineering Molecules to Materials
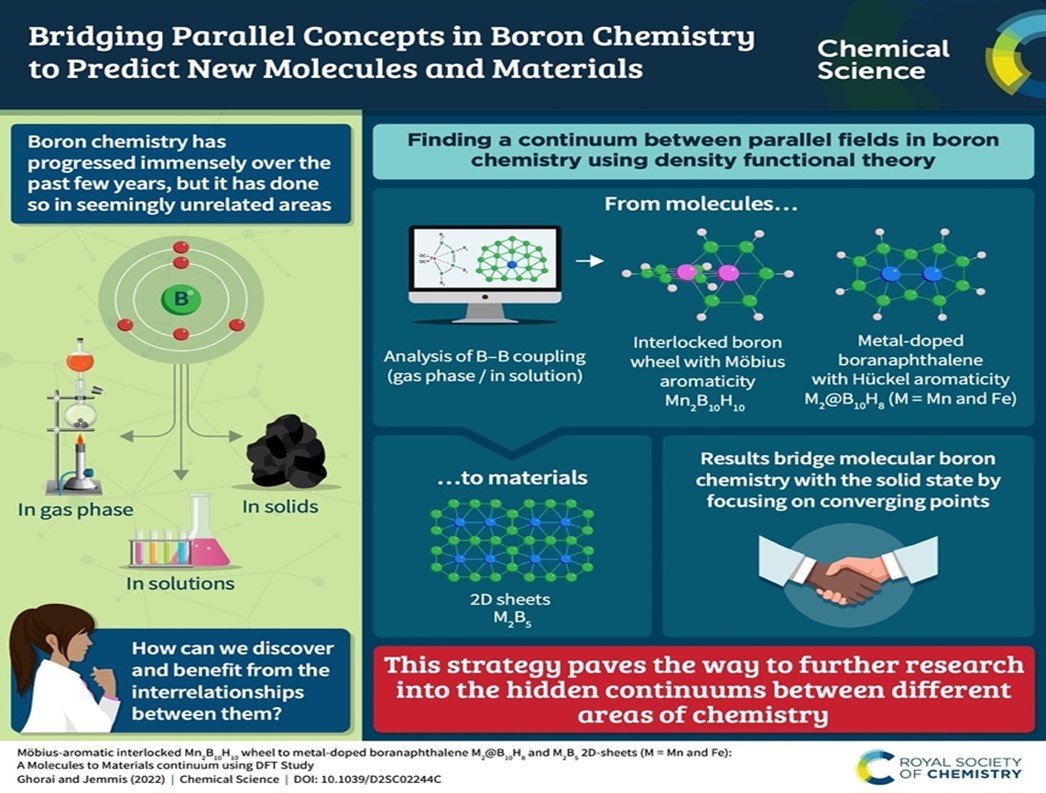
Chemistry takes place in solution, in the gas phase, in the solid. In recent years, chemistry of boron has developed in unimaginable ways in each of these phases. Researchers in the Department of Inorganic and Physical Chemistry led by E D Jemmis use quantum mechanics-based electronic structure theory to understand the molecular chemistry in the gas phase and in solution, and apply the information as a design element to predict 2D materials – a process they term as Orbital Engineering.
For a casual observer the new developments in the chemistry of boron in solution, in the gas phase, and in the solid appear as unrelated. The researchers imagine them as points along a continuum in the larger canvas of molecules-clusters-solids. The interrelationships among them help to devise a molecules-to-materials strategy starting from a series of novel boron compounds with unusual bonding possibilities.
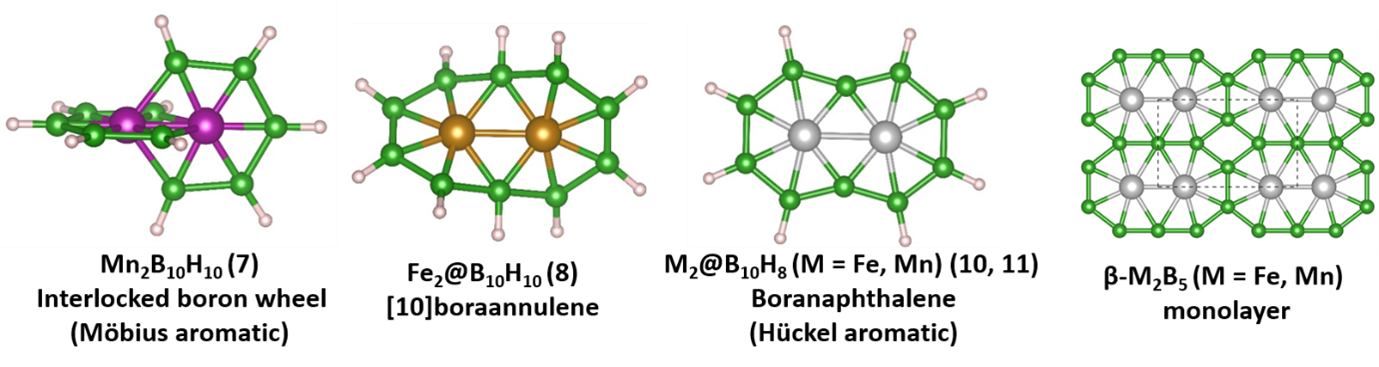
In a new study, the team merged the B-B bond forming reactions of Himmel, Kinjo, Braunschweig and others in solution, and the gas phase metal boron clusters of Wang, to predict an interlocked molecular boron wheel Mn2B10H10 having two intervening rings orthogonal to each other. This is predicted to have 8π Möbius aromaticity. Orbital Engineering helped them to add two electrons to boron wheel Mn2B10H10 by replacing Fe by Mn, making the structure near planar. Further transformation to planar Hückel 10π-aromatic boranaphthalene M2@B10H8 (M = Mn or Fe) was easy. Finally, just as poly-condensation of benzene or naphthalene leads to 2D graphene, M2@B10H8 could give metal boride monolayers, M2B5, considering M2@B10 as the building block (M = Mn and Fe). Molecular boron chemistry and 2D-metal borides form a part of the boron chemistry continuum. This approach lends itself to obvious extensions.



Reference:
This research is published in Chemical Science, the Royal Society of Chemistry’s peer-reviewed flagship journal, and is free to read (https://pubs.rsc.org/en/content/articlelanding/2022/sc/d2sc02244c)
Website URL: https://ipc.iisc.ac.in/~edj/








