Understanding metastasis in cancer to fight it better
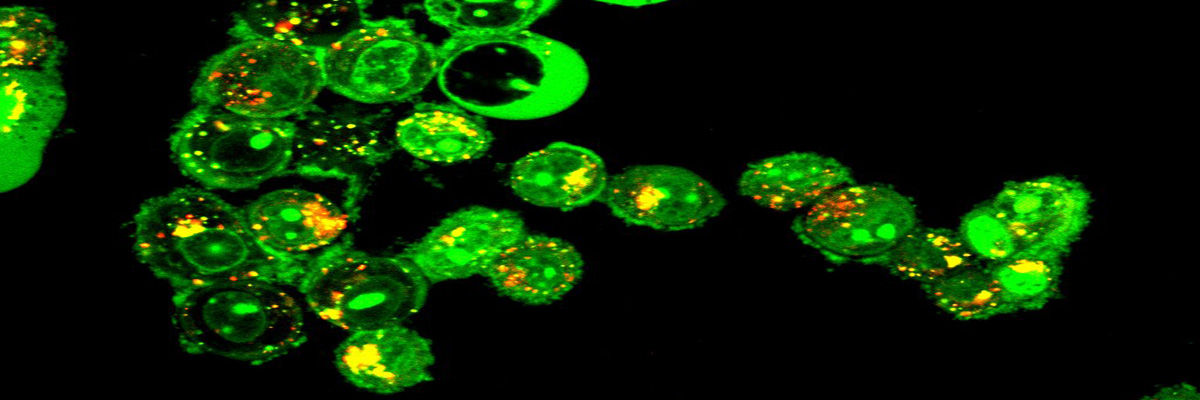
Confocal image of acridine orange stained acidic vesicles (red/orange puncta) in matrix-detached MCF7 breast cancer cells showing autophagy.(Figure: Courtesy Saurav Kumar Ph.D. student AR lab;)
Cancer, a dreaded disease, has been on a rampant spread over the past decade. Reports from the Indian Council of Medical Research (ICMR) estimated 14.5 lakh new cancer cases in 2016, and this number is likely to go up to 17.3 lakhs in 2020. Scientists around the world are uncovering new facets of this disease in a bid to understand its many faces and design the best and the most effective drugs against it. So far, we know that abnormal and uncontrollable growth of cells in our body leads to cancer. In advanced stages, these cancerous cells spread to different parts of the body through our blood or lymph, in a process called metastasis. In a new study, researchers from the Indian Institute of Science, Bengaluru, led by Prof. Annapoorni Rangarajan, have made some astounding discoveries on the molecular mechanism behind the spread of cancer, which can help better understand and treat the disease.
But how exactly does metastasis happen? Cancerous cells from a tumour, called ‘circulating tumour cells’ or CTCs, enter the bloodstream or the lymph system and move throughout the body. With the help of proteins called integrins, these tumour cells adhere to the extracellular matrix – a collection of molecules that support the cell structure. Once they find a suitable place, they thrive and develop into another cancerous tumour. However, if these tumour cells are deprived of the matrix, they die a form of programmed cell death called anoikis. For cancer to thrive, the tumour cells must develop resistance to anoikis. But how do they develop resistance? That is what the researchers of this study have uncovered.
The scientists studied the roles of Akt – an enzyme that regulates the process of survival and metabolism in a cell, and AMP activated protein kinase (AMPK) – an enzyme that promotes the breakdown of complex molecules into simpler ones for the cell to utilise. They used human breast cancer cells and mice for the study. While Akt drives cell growth and multiplication, thus promoting tumor growth, AMPK was long thought of as a tumor suppressor because of its growth retarding effects. But, recent studies have shed light on how AMPK could also aid tumour growth, as it helps the cell survive when it is in need of energy or in an environment of reduced oxygen.
“It was about ten years ago, when a graduate student in the lab, Sravanth, made the first observation that AMPK is activated upon matrix-detachment”, reveals Prof. Rangarajan about the motivation behind this study. This surprising observation made the team question the role of AMPK in resisting anoikis, while most of the studies until then had always credited Akt for this phenomenon. “We were inquisitive to check the status of Akt in detached cells, and to our surprise, we found that Akt is markedly inactivated in detached cells”, she adds.
The researchers of the study observed that AMPK is activated in detached cells compared to adherent cells, whereas Akt is more activate in adherent cells compared to detached. When tumor cells detach from their primary site, they are marked for death. This is because they are anchorage-dependent cells and need to be attached to the extracellular matrix to grow and divide. When detached, they trigger the enzyme AMPK, which represses the enzyme Akt by a protein called PHLPP2. When the tumour cells re-attach to the matrix, the Akt enzyme is triggered. This represses AMPK via another protein called PP2Ca, therefore showing a double negative feedback loop that helps the tumor cells to adapt to matrix deprivation.
“Our study suggests that AMPK inhibition will help prevent cancer spread by metastasis. Currently, metformin, a first line anti-diabetic drug that ironically activates AMPK is being trialled in cancer treatment. Our study raises concerns on the use of AMPK-modulating agents before further detailed investigations. A side effect of metformin might actually promote cancer spread in some cases”, says Prof. Rangarajan about the study.
This study provides newer insights into developing AMPK and PHLPP2 inhibitors as potential drugs against cancer that can target the molecular mechanisms involved in the spread of cancer. As a next step, the researchers are pursuing studies to understand this complex mechanism better. “We have showed that AMPK activation upregulates the phosphatase PHLPP2 to inhibit Akt. However, the mechanism(s) by which AMPK does this is not known. We are currently pursuing this”, says Prof. Rangarajan, before signing off.
| Author(s) of research paper | Manipa Saha¶, Saurav Kumar¶, Shoiab Bukhari, Sai A Balaji, Prashant Kumar†, Sravanth K, Hindupur, and Annapoorni Rangarajan
Department of Molecular Reproduction, Development and Genetics, Indian Institute of Science
†Institute of Bioinformatics
|
| Title of research | AMPK-AKT double negative feedback loop in breast cancer cells regulates their adaptation to matrix deprivation |
| Funding Information | This work was supported by the Wellcome Trust/DBT India Alliance Fellowship |
| Article written by | Article written by Annie Megan Santamaria |
Reference: http://cancerres.aacrjournals.org/content/early/2018/01/13/0008-5472.CAN-17-2090
Other Featured research




